Protecting mothers and babies from HIV and syphilis as price of the dual test falls to $0.84
How did a unique partnership increase access to syphilis testing for millions of women while saving millions for countries and procurers?
23 January 2026 | Insight
Testing pregnant women for HIV and syphilis is vital for the protection of mothers and babies. Although testing rates for HIV have reached historic highs in many countries, testing for syphilis – a leading cause of stillbirths and newborn deaths – has historically lagged, with devastating consequences.
Effective point-of-care testing tools for syphilis were available thanks to scientific innovation. But these tests were not reaching antenatal clinics in low- and middle-income countries. A mix of market barriers, misaligned incentives and weak delivery models led to a yawning syphilis testing gap, leaving millions of women, and their unborn and newborn babies unreached.
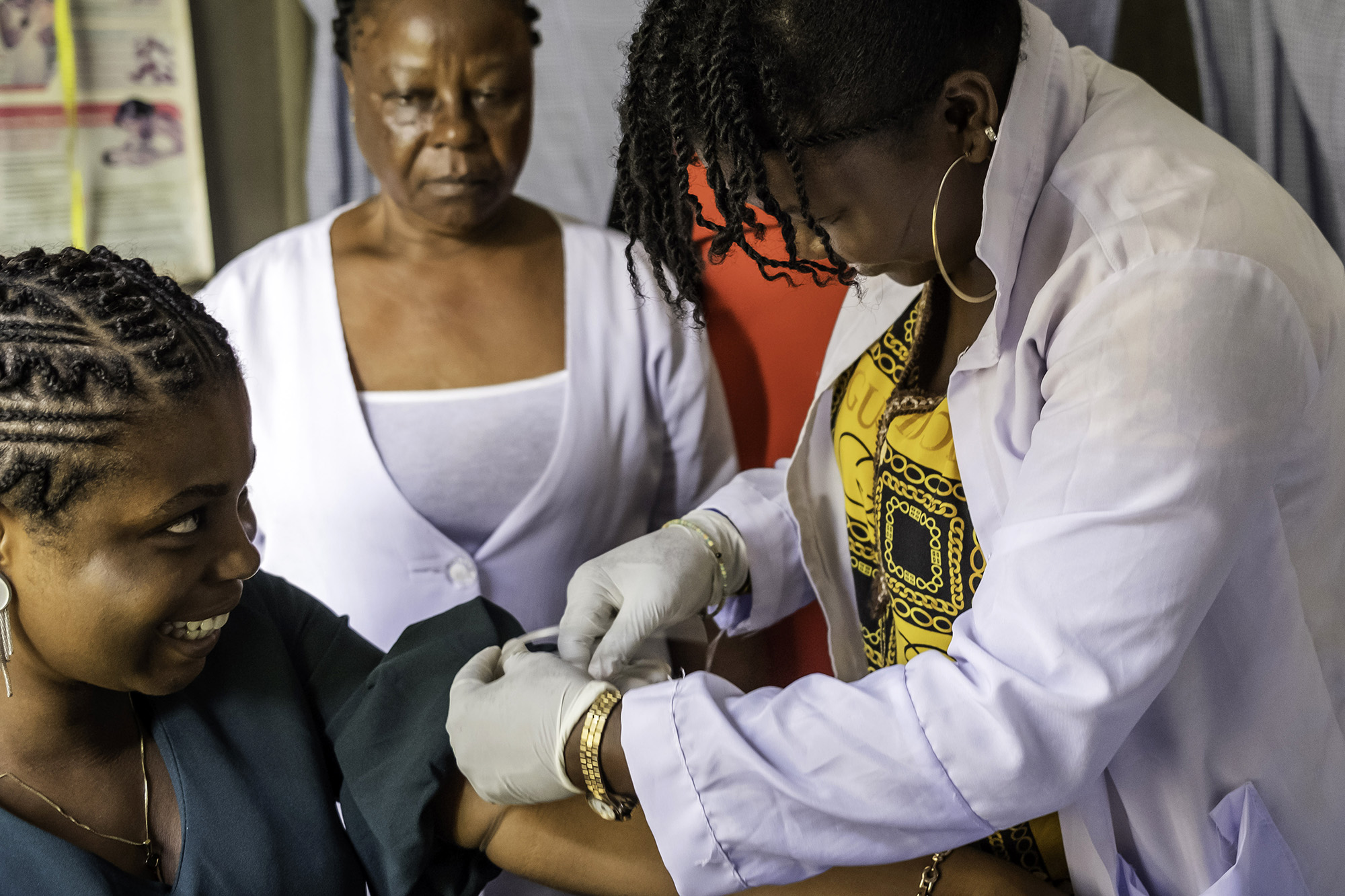
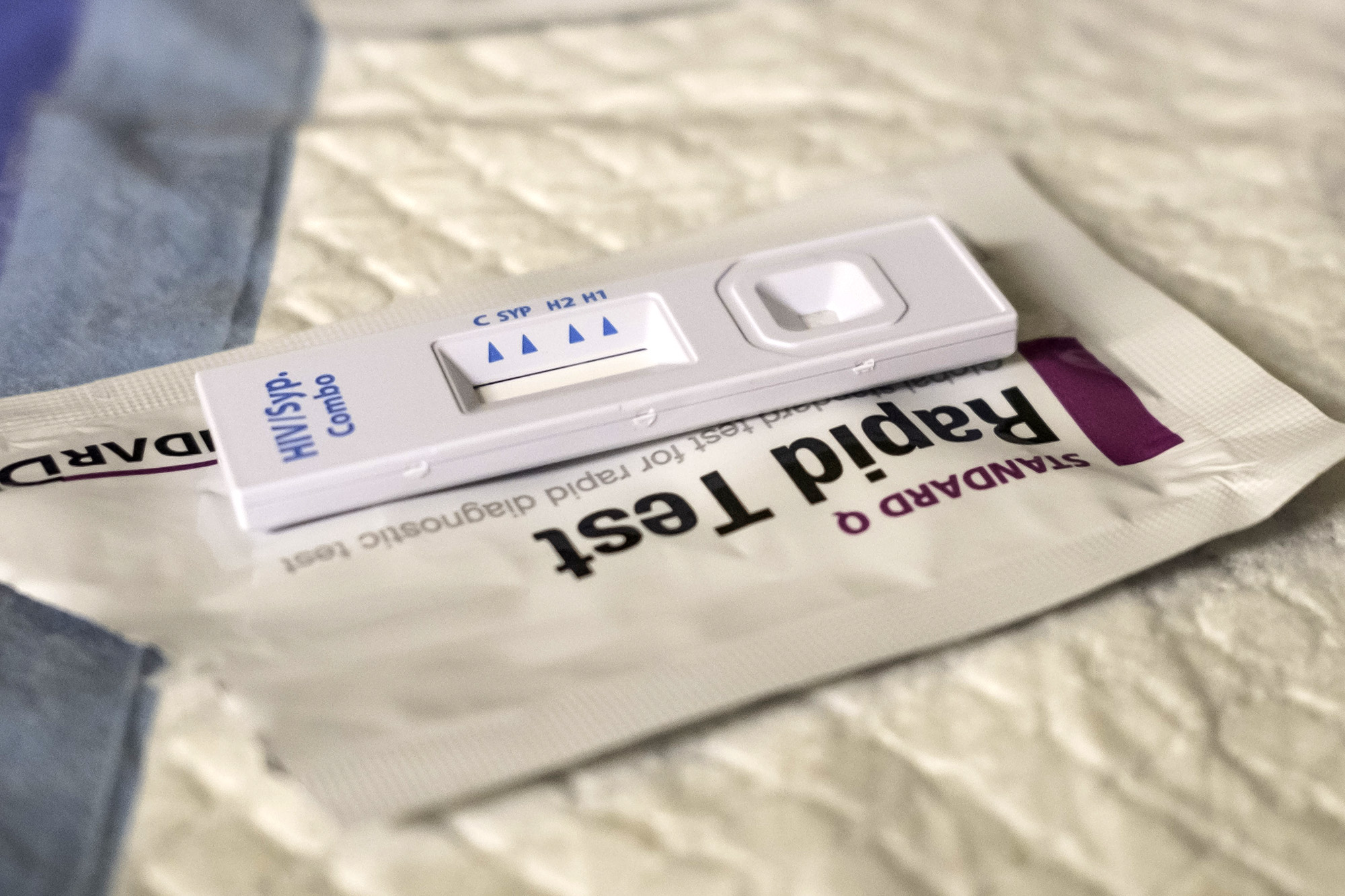
Today, countries can purchase an innovative dual test for HIV and syphilis for a record low price of $0.84. The test, administered by healthcare workers at clinics or in homes, provides a result within 20 minutes. Women who find out they have HIV can begin antiretroviral treatment whilst those diagnosed with syphilis can be treated with an inexpensive antibiotic.
This record low price did not happen by accident. It was the result of a unique partnership between MedAccess, the Clinton Health Access Initiative and the manufacturer, SD Biosensor that catalysed the market and continues to expand access to HIV/syphilis dual tests today. SD Biosensor has now announced that its dual HIV/syphilis test is available at $0.84 ex works for eligible low- and middle-income countries under the MedAccess–CHAI partnership, a significant reduction from the price in the original agreement of just under $1. This reflects rapid growth in the market and opens new possibilities for countries to expand testing within even tighter budget constraints.
Here we hear from five of the people involved in efforts to improve access to HIV and syphilis testing for millions of mothers.
The context
Andrew Storey, Director, Sustainable Markets, Impact Nexus Africa
Before 2020, the global response to congenital syphilis was defined by a persistent implementation gap: tests and treatments to prevent transmission existed, but they were not reaching women at scale. A 2019 Lancet Global Health paper set out the core hypothesis that market barriers – such as incomplete demand forecasts and inefficient distribution of syphilis tests – rather than clinical uncertainty, were leading to low syphilis testing rates. At the time, an estimated 930,000 pregnant women were infected with syphilis each year, resulting in more than 350,000 adverse birth outcomes, including stillbirth, neonatal death, and severe disability. These outcomes were largely preventable with timely testing and treatment.

Yet in many high-burden countries across Africa and South Asia, fewer than half of pregnant women were tested for syphilis in antenatal care, even where HIV testing coverage exceeded 80 per cent. Syphilis testing lagged because it sat in a structural blind spot: procurement was fragmented into small, uncoordinated volumes, there was no strong single-disease political or financing constituency, and providing separate tests for HIV and syphilis added operational burden for frontline staff, increasing the risk that syphilis was simply deprioritised in busy clinics.
Dual HIV/syphilis rapid diagnostic tests (RDTs) emerged in this context not just as a new innovation, but as a systems solution that collapsed two pathways into one, reduced clinic-level workload, and created the conditions for a more coherent and sustainable market for syphilis testing.
The market
Hema Srinivasan, Senior Adviser, Impact Nexus Africa
Colin Brown, Senior Advisor, Global Markets Team, CHAI
By 2020, leading diagnostic manufacturers like Abbott, Premier, and SD Biosensor had developed dual HIV/syphilis RDTs – simple, lateral flow assays that could test for both HIV and syphilis infection with a single drop of blood.
The dual HIV/syphilis RDT provided a unique opportunity to leverage robust HIV testing programs to increase syphilis testing coverage, simply by replacing an HIV test with one that tested for both HIV and syphilis at the same time.

But donors wanted to rationalise spending on HIV rapid tests, given the growing need for treatments and prevention tools, so budgets were tight. While dual testing was technically cost-effective at the prevailing $1.50 price in 2019, public health leaders in sub-Saharan Africa called for a sub-$1 price. This would still be higher than the $0.70-0.80 price of single HIV tests but would enable rapid scale-up of dual RDTs within constrained budget envelopes.
For suppliers, committing to a $1 price point would be a risky move. Near-term profit margins would drop to almost nothing if production volumes got stuck at less than 1 million tests per year. But if the lower price point enabled rapid expansion in syphilis testing coverage among pregnant women and other key populations, then the market could grow to 20-30 million tests within a few years, and all suppliers could sustain their commercial operations at a sub-$1 price.
CHAI and MedAccess approached all three WHO-prequalified suppliers to present this market vision and offer a multi-year volume guarantee that would de-risk the decision to dramatically cut prices of the dual RDT. SD Biosensor was excited by the opportunity to help expand access to syphilis and HIV testing and agreed to offer a sub-$1 price for the dual RDT across all low- and middle-income countries.
The guarantee and implementation support
In November 2021, MedAccess, CHAI and SD Biosensor announced a partnership to reduce the price of the company’s dual test to under $1 in more than 100 low- and-middle income countries. This initial commitment laid the foundation for later reductions, with the price now falling further to $0.84 as demand expanded and production scaled.
MedAccess provided a volume guarantee to SD Biosensor, committing to make a shortfall payment to the manufacturer if guaranteed sales volumes were not achieved. This transferred early market entry risk away from SD Biosensor and made it possible to reduce the price of its STANDARD Q HIV/Syphilis Combo test by 32 per cent.
To boost the success of the partnership, MedAccess worked with CHAI to provide active implementation support to SD Biosensor and countries. This implementation support led to smoother adoption of new policies by countries, coordination of healthcare worker trainings and translation of demand to actual testing. As a result, procurement across supported countries became routine, to the benefit of the healthcare system.
The impact
Dr Tristana Perez, Head of Impact, MedAccess
Taking the risk paid off. The new price point made dual RDTs an attractive option for countries aiming to reach more pregnant women and prevent stillbirths and newborn deaths, exceeding all of our projections. We’ve seen rapid expansion and purchases of these tests at sub-$1 prices across more than 60 low- and middle-income countries by the end of 2024 – many of them smaller nations who had not previously planned to introduce the tests.
Millions of women have gained access to a key element of maternal care and devastating outcomes for babies have been averted. As of the end of 2024, an estimated 31 million pregnant women accessed dual tests under guarantee terms, resulting in more than 90,000 stillbirths being averted thanks to timely treatment with low-cost, widely available antibiotics.

Countries have also benefited financially. The reduction of average market prices by 21% also delivered $11.3 million in direct savings to procurers, including ministries of health using domestic budgets. Beyond this, there are wider savings to the health system from preventing ill health.
With implementation support from CHAI in 14 priority countries, governments have expanded procurement and accelerated rollout. For example, from the start of our agreement to the end of 2024, over 39,000 antenatal clinics in Nigeria started offering the dual test, increasing syphilis testing rates from 24 per cent to 58 per cent. With CHAI’s support, our guarantee has helped to build a diverse and sustainable market for dual testing.
The latest drop to $0.84 represents a new phase of the market’s evolution, showing how early risk-sharing and coordinated demand can continue to drive prices down over time, even after the original guarantee period.
The future
Victoria Goodfellow, Head of Infectious Diseases, MedAccess
The dramatic price reductions achieved for dual rapid diagnostic tests have delivered substantial public health value. Reaching price stability at this level can now serve as a source of resilience for both buyers and suppliers, enabling governments and other procurers to plan and budget with greater confidence and supporting a gradual transition to domestic financing. At the same time, it is important to signal the need for price stabilisation for manufacturers in order to protect longer-term market health and, ultimately, patient access. Experience from other segments of the RDT market, most notably malaria, shows that a race to the bottom on price can erode margins to the point that risks supplier exits, weakening competition and undermining access objectives.
Ongoing constraints in the funding environment mean that trade-offs are unavoidable and efficiency matters more than ever. Integrated dual testing delivers strong value by simplifying testing algorithms, lowering service delivery costs, and maximising the impact of each patient interaction. Preserving a healthy, competitive dual RDT market is therefore critical to maintaining coverage gains during this period of funding transition.

Looking ahead, triple HIV, syphilis, and hepatitis B RDTs represent the next major opportunity. The combination of three tests could bring hepatitis B screening to millions who are currently missed, particularly in antenatal care and among key populations. The lessons from dual RDTs are clear. Early coordination, credible demand signals, regulatory alignment, and shared risk can change what looks commercially impossible into something routine. With the right conditions and support in place, the triple RDTs could follow the same path, offering a compelling route to expanded diagnostic coverage and more efficient, accessible testing.
We are at an exciting moment. In the coming years, we could see millions more people, including those from key populations, tested for potentially fatal illnesses. But this is, of course, only one part of the story. As availability of dual and triple RDTs increases, this needs to be matched by access to complementary tests, treatments and other consumables. Partnerships like the one between MedAccess, CHAI and SD Biosensor offer a compelling roadmap for increasing access to lifesaving tools.
What’s next?
In late January 2026, Governments, suppliers, and global partners will meet in Johannesburg at a leadership meeting organised by Impact Nexus Africa, with the support of the Gates Foundation and South Africa’s National Department of Health. The focus will be simple: how to sustain access to dual RDTs, and how to build the conditions for the next generation of integrated diagnostics during a period of tight financing and programmatic transition.
Acknowledgements
Increasing access to essential medical products is a collective effort, with national governments and local health systems playing the central role in scaling up use of Dual RDTs in antenatal care globally. Alongside recognising the leadership of countries themselves and the tireless work of those delivering these programmes, the authors would also like to acknowledge our partners for their contributions to expanding access to HIV and syphilis testing. These include Evidence Action; Foreign, Commonwealth & Development Office; Gates Foundation; Global Fund for AIDS, Tuberculosis and Malaria; Healthy Futures Global; Path; Pepfar; Solthis; Unitaid; and the World Health Organization.
About the authors
Andrew Storey was Senior Director, Maternal and Neonatal Health at CHAI between 2018 and 2025.
Hema Srinvasan was Chief Access Officer at MedAccess between 2020 and 2023.
Colin Brown is Senior Advisor, Global Health Markets Team at CHAI.
Dr Tristana Perez is Head of Impact at MedAccess.
Victoria Goodfellow is Head of Infectious Diseases at MedAccess.